Accelerate Study Start-up. Activate Sites Faster. Launch With Confidence.
Cloudbyz Study Start-up turns feasibility, regulatory, contracts, vendor readiness, and site activation into one connected workflow — so clinical teams reduce cycle time, eliminate delays, and stay inspection-ready from day one.
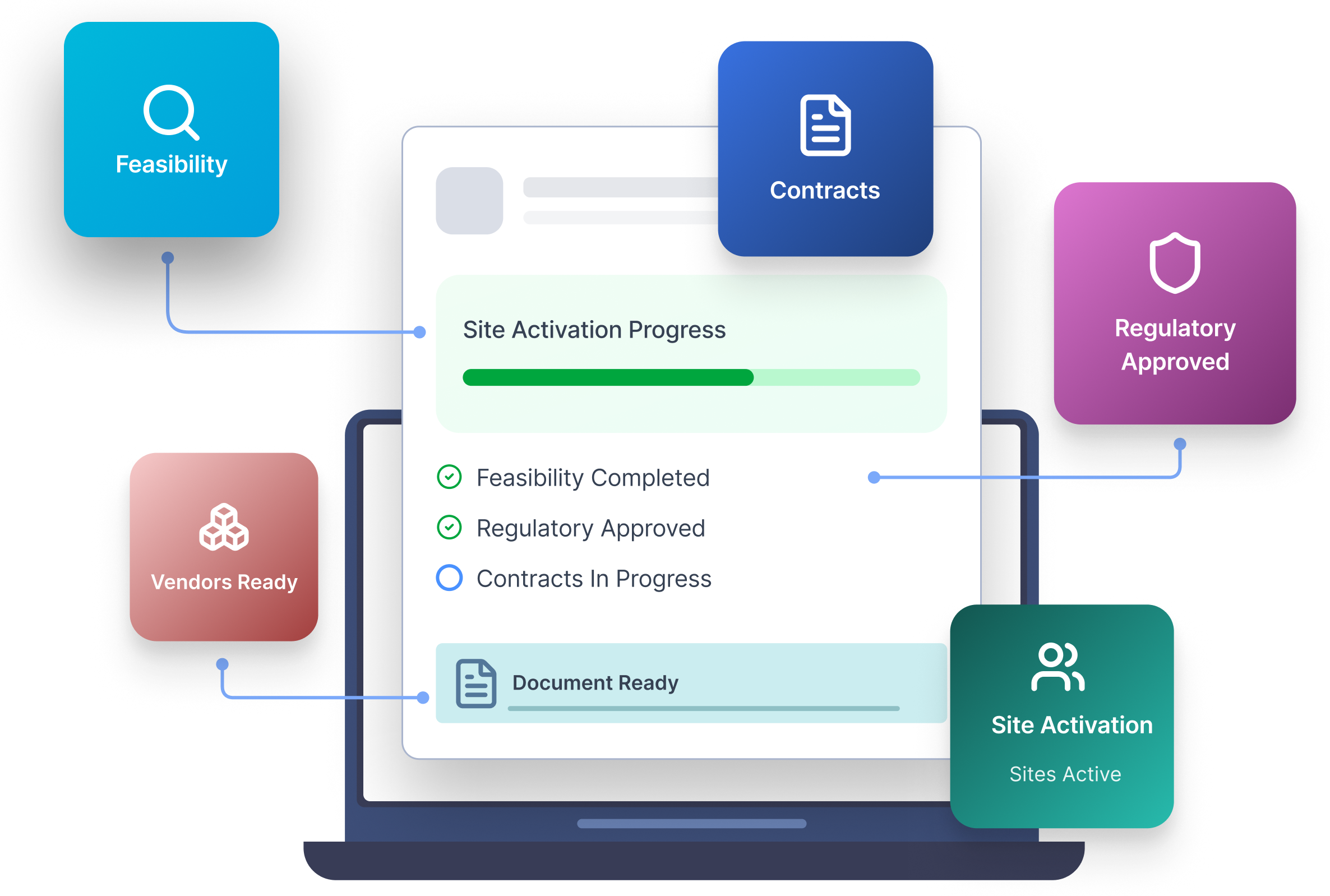
Study Start-up Is Still Where Clinical Operations Loses Time and Control

What Cloudbyz Study Start-up Delivers

Faster site activation and reduced startup cycle time

Real-time visibility across countries, sites, and vendors

Fewer handoff errors and less rework

Better predictability for enrollment and timelines

Stronger inspection readiness through evidence-backed execution

Scale start-up without scaling headcount

More Than Tracking — Study Start-up as an Execution System
Most tools track start-up status. Cloudbyz executes it.
- Cloudbyz Study Start-up standardizes key startup milestones to ensure consistent and structured execution across studies.
- It automates workflows to streamline processes and reduce manual coordination efforts.
- The system captures and maintains documented evidence to support readiness and compliance.
- Fully unified with Cloudbyz CTMS and the broader Cloudbyz eClinical platform, it creates a single source of truth for study readiness.
Core Capabilities
Milestone Packs, Dependencies & Readiness Scoring
Standardize What “Ready” Means — Across Every Country and Site
Cloudbyz replaces inconsistent startup trackers with structured milestone packs aligned to your SOPs. Teams can define readiness once, reuse it across studies, and scale globally without reinventing the process.
Includes:
- Country and site milestone templates
- Dependency logic (parallel vs blocking tasks)
- Readiness scoring (Green / Yellow / Red)
- Portfolio dashboards across studies, countries, and sites
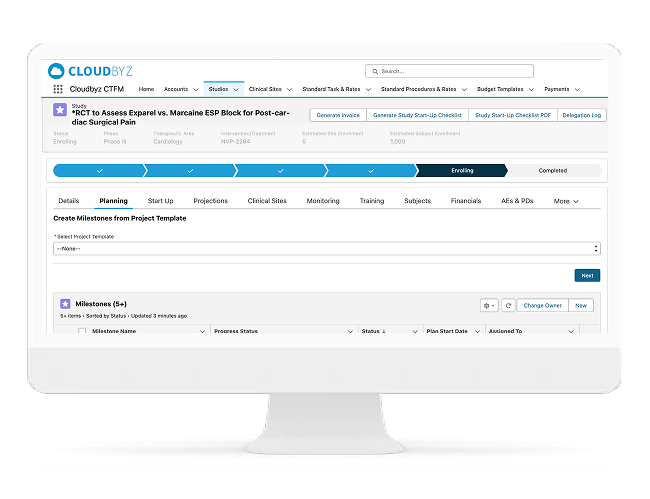
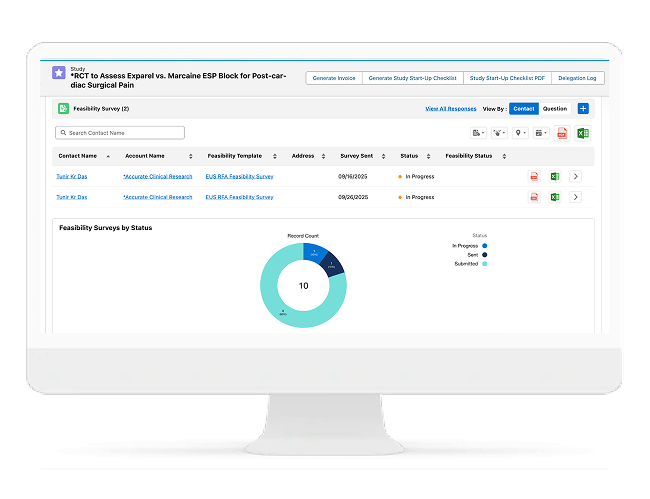
Site Feasibility, Profiling & Selection
Faster Feasibility With Data-Driven Site Selection
Cloudbyz enables sponsors and CROs to run feasibility and site profiling at scale. Capture structured responses, compare sites consistently, and make faster selection decisions with confidence.
Includes:
- Configurable feasibility questionnaires
- Site profiling and capabilities tracking
- Site scoring models (experience, capacity, performance)
- Historical site performance and reuse of preferred sites
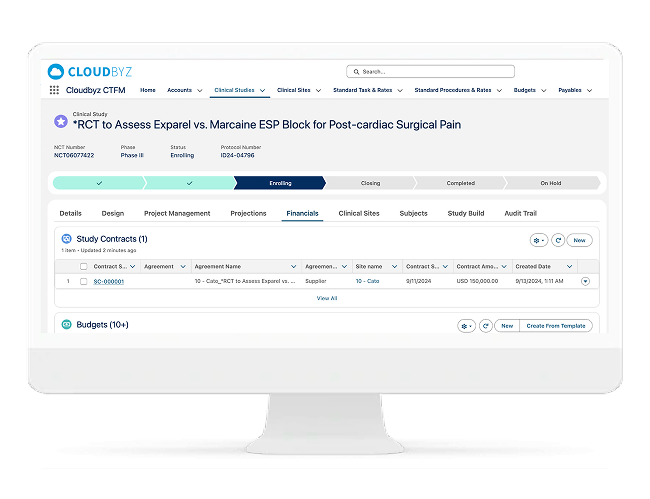
Contract & Budget Workflow Tracking
Make Contracts and Budgets Visible — Without Slowing the Team
Contract and budget negotiation is one of the biggest sources of activation delays. Cloudbyz brings CTA and budget workflows into the start-up execution layer so teams can see bottlenecks early and keep timelines realistic.
Includes:
- CTA and budget status workflows
- Redline and negotiation cycle tracking
- Approvals, escalations, and audit history
- Capture key terms and payment triggers
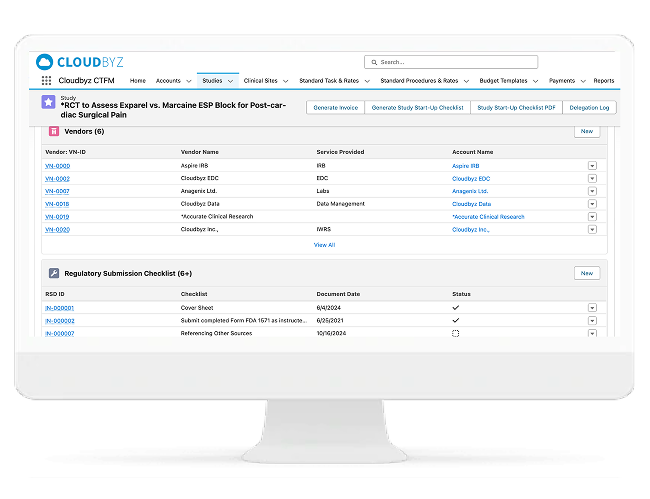
Regulatory Submission & Approval Tracking
Manage Regulatory and Ethics Readiness as a Controlled Process
Cloudbyz centralizes IRB/EC submissions and regulatory approvals so teams always know what has been submitted, what is approved, what is expiring, and what is blocking activation.
Includes:
- Central vs local IRB/EC tracking
- Country regulatory authority submissions
- Approval expiry, renewal, and amendment tracking
- Submission completeness checks and evidence capture

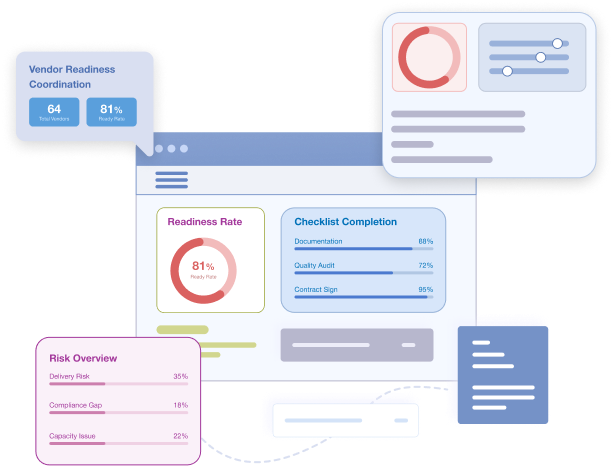
Vendor Readiness Coordination
Bring Vendor Start-up Into One Control Plane
Start-up timelines are often delayed by vendors: labs, imaging, IRT, logistics, and eCOA. Cloudbyz tracks vendor deliverables in the same readiness model so you can coordinate across all parties with shared accountability.
Includes:
- Vendor onboarding checklists and milestones
- Lab, imaging, IRT/RTSM, eCOA, and logistics readiness
- Shared views across sponsors, CROs, and vendors
- Delay tracking and escalation workflows
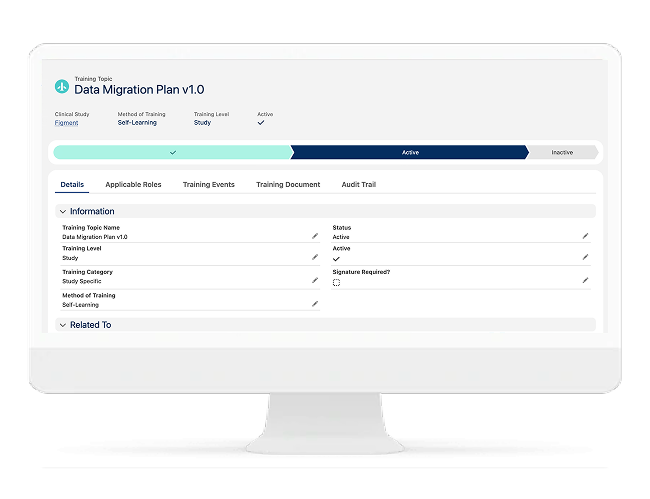
Site Onboarding & Training Readiness
Ensure Sites Are Truly Ready to Enroll
A site is not ready because an approval exists — it is ready when staff, training, systems access, and documentation are complete. Cloudbyz helps teams standardize onboarding and capture readiness evidence.
Includes:
- Site training checklists (protocol, GCP, EDC, IRT, safety)
- Training completion evidence capture
- Staff roster and role readiness
- Activation gating logic based on readiness
Essential Documents & Inspection Readiness
Build Inspection Readiness Into Start-up — Not After
Cloudbyz captures evidence as part of the workflow. Every milestone can require documentation, approvals, and artifacts — automatically organized and audit-ready.
Includes:
- Evidence requirements per milestone
- Document versioning and audit trails
- “Proof of readiness” summaries by site/country
- Continuous readiness instead of last-minute cleanup
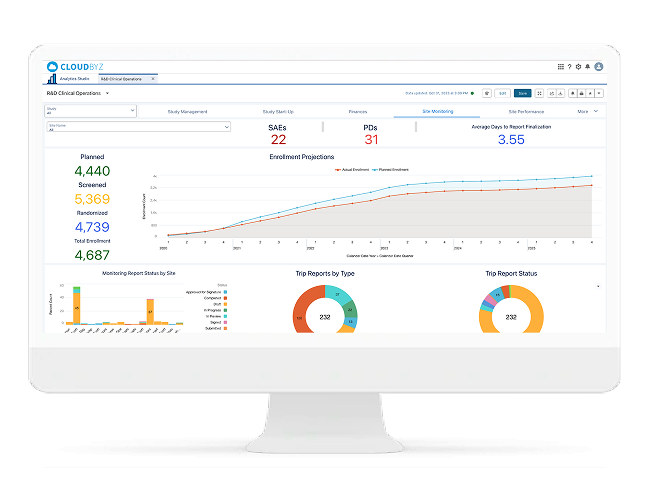
Activation Forecasting & Bottleneck Analytics
Predict Activation — Don’t Just Report Status
Cloudbyz turns start-up execution data into forecasting and analytics. Teams can see expected activation timelines, top delay drivers, and where intervention will make the biggest impact.
Includes:
- Forecasted activation dates by site/country
- Cycle-time benchmarking
- Bottleneck dashboards (contracts, regulatory, vendors, training)
- Executive-ready weekly summaries
AI Agents for Study Start-up
AI Agents That Remove the Manual Work — Without Losing Control
Study start-up is high-volume, document-heavy, and full of repetitive follow-ups. Cloudbyz AI Agents automate the work that slows teams down while maintaining audit trails and human oversight.
AI Feasibility Agent

AI Start-up Coordinator Agent

AI eTMF Agent

AI Executive Insights Agent

Built for Trust and Compliance
Human-in-the-loop approvals
Full audit trail of AI actions and recommendations
Configurable rules aligned to SOPs
Secure governance aligned to GxP expectations
Why Cloudbyz Study Start-up Is Different

Unified With CTMS and the eClinical Platform
Cloudbyz connects start-up workflows directly to CTMS execution, eTMF evidence, and downstream financial processes — eliminating handoff gaps.

Designed for Execution, Not Just Tracking
Milestone packs, dependencies, gating, evidence, and automation create a system that drives work forward.

Built on Salesforce
Enterprise-grade scalability, security, and global performance — with configuration flexibility for complex organizations.

Configurable to Your SOPs
No rigid templates. Configure start-up workflows to match your organization's processes and governance.
Built for the Teams Who Own Start-up
Sponsors
Standardize global start-up, reduce cycle time, and improve predictability.
CROs
Run multiple studies in parallel with consistent execution and shared accountability.
Site Networks
Streamline onboarding, training, documentation, and activation readiness.
Enterprise-Grade Security and Auditability





















